Kolekce Jj Thomson Experiment Atom Výborně
Kolekce Jj Thomson Experiment Atom Výborně. Thomson, a british physicist, conducted the cathode ray experiment. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.
Tady What Is Jj Thomson S Plum Pudding Model
Thomson, a british physicist, conducted the cathode ray experiment. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Previously, atoms were known to be indivisible, but in 1897, j. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.He zapped atoms with electricity and observed that negatively charged particles were removed!
He zapped atoms with electricity and observed that negatively charged particles were removed! An atom resembles a sphere of positive … Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. He zapped atoms with electricity and observed that negatively charged particles were removed! It is a vacuum sealed tube with a cathode and anode on one side. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He did this using a cathode ray tube or crt.

Previously, atoms were known to be indivisible, but in 1897, j... An atom resembles a sphere of positive … He did this using a cathode ray tube or crt. Thomson, a british physicist, conducted the cathode ray experiment. Previously, atoms were known to be indivisible, but in 1897, j. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. It is a vacuum sealed tube with a cathode and anode on one side. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons... Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.

He did this using a cathode ray tube or crt. .. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle... He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.

He did this using a cathode ray tube or crt... Thomson, a british physicist, conducted the cathode ray experiment. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Previously, atoms were known to be indivisible, but in 1897, j. It is a vacuum sealed tube with a cathode and anode on one side. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. An atom resembles a sphere of positive … He zapped atoms with electricity and observed that negatively charged particles were removed! After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He did this using a cathode ray tube or crt.. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

Thomson, a british physicist, conducted the cathode ray experiment. Thomson, a british physicist, conducted the cathode ray experiment. It is a vacuum sealed tube with a cathode and anode on one side. He zapped atoms with electricity and observed that negatively charged particles were removed! He did this using a cathode ray tube or crt. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. An atom resembles a sphere of positive … He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He did this using a cathode ray tube or crt.

Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Previously, atoms were known to be indivisible, but in 1897, j. He zapped atoms with electricity and observed that negatively charged particles were removed! An atom resembles a sphere of positive … It is a vacuum sealed tube with a cathode and anode on one side. He did this using a cathode ray tube or crt.

It is a vacuum sealed tube with a cathode and anode on one side.. He did this using a cathode ray tube or crt. Previously, atoms were known to be indivisible, but in 1897, j.. Previously, atoms were known to be indivisible, but in 1897, j.

He zapped atoms with electricity and observed that negatively charged particles were removed! .. He did this using a cathode ray tube or crt.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them... He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. It is a vacuum sealed tube with a cathode and anode on one side. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Previously, atoms were known to be indivisible, but in 1897, j. An atom resembles a sphere of positive ….. It is a vacuum sealed tube with a cathode and anode on one side.

An atom resembles a sphere of positive ….. He did this using a cathode ray tube or crt. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. An atom resembles a sphere of positive … Thomson, a british physicist, conducted the cathode ray experiment. Previously, atoms were known to be indivisible, but in 1897, j. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He zapped atoms with electricity and observed that negatively charged particles were removed! It is a vacuum sealed tube with a cathode and anode on one side. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.
It is a vacuum sealed tube with a cathode and anode on one side... An atom resembles a sphere of positive … He zapped atoms with electricity and observed that negatively charged particles were removed! He did this using a cathode ray tube or crt. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson, a british physicist, conducted the cathode ray experiment. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.
Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.. He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He did this using a cathode ray tube or crt. It is a vacuum sealed tube with a cathode and anode on one side.. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

An atom resembles a sphere of positive … He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson, a british physicist, conducted the cathode ray experiment. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. An atom resembles a sphere of positive … An atom resembles a sphere of positive …

He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Previously, atoms were known to be indivisible, but in 1897, j. He did this using a cathode ray tube or crt.

Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons... He zapped atoms with electricity and observed that negatively charged particles were removed! An atom resembles a sphere of positive … He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He did this using a cathode ray tube or crt. Previously, atoms were known to be indivisible, but in 1897, j.. It is a vacuum sealed tube with a cathode and anode on one side.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He did this using a cathode ray tube or crt. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson, a british physicist, conducted the cathode ray experiment. An atom resembles a sphere of positive … After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. It is a vacuum sealed tube with a cathode and anode on one side. Previously, atoms were known to be indivisible, but in 1897, j... It is a vacuum sealed tube with a cathode and anode on one side.

Previously, atoms were known to be indivisible, but in 1897, j. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Previously, atoms were known to be indivisible, but in 1897, j.

Thomson, a british physicist, conducted the cathode ray experiment.. Previously, atoms were known to be indivisible, but in 1897, j. It is a vacuum sealed tube with a cathode and anode on one side. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.. An atom resembles a sphere of positive …

Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. An atom resembles a sphere of positive ….. Previously, atoms were known to be indivisible, but in 1897, j.
.PNG)
He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles... After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson, a british physicist, conducted the cathode ray experiment. It is a vacuum sealed tube with a cathode and anode on one side... Thomson, a british physicist, conducted the cathode ray experiment.

He did this using a cathode ray tube or crt. Thomson, a british physicist, conducted the cathode ray experiment. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. It is a vacuum sealed tube with a cathode and anode on one side. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

An atom resembles a sphere of positive … He zapped atoms with electricity and observed that negatively charged particles were removed! An atom resembles a sphere of positive … It is a vacuum sealed tube with a cathode and anode on one side. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.
Previously, atoms were known to be indivisible, but in 1897, j. Previously, atoms were known to be indivisible, but in 1897, j. Thomson, a british physicist, conducted the cathode ray experiment. An atom resembles a sphere of positive … He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He did this using a cathode ray tube or crt. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. It is a vacuum sealed tube with a cathode and anode on one side. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson, a british physicist, conducted the cathode ray experiment.

Thomson, a british physicist, conducted the cathode ray experiment... After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. An atom resembles a sphere of positive … It is a vacuum sealed tube with a cathode and anode on one side. Thomson, a british physicist, conducted the cathode ray experiment.

He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

Previously, atoms were known to be indivisible, but in 1897, j. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Previously, atoms were known to be indivisible, but in 1897, j. He zapped atoms with electricity and observed that negatively charged particles were removed!. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.
After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He zapped atoms with electricity and observed that negatively charged particles were removed!
.PNG)
He did this using a cathode ray tube or crt.. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He did this using a cathode ray tube or crt. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson, a british physicist, conducted the cathode ray experiment. An atom resembles a sphere of positive … Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He zapped atoms with electricity and observed that negatively charged particles were removed!
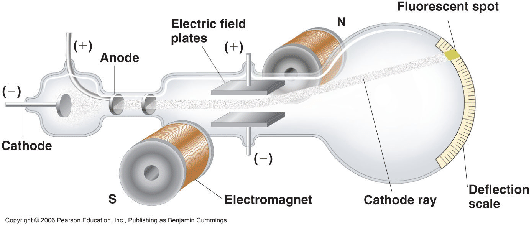
He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. It is a vacuum sealed tube with a cathode and anode on one side. An atom resembles a sphere of positive …

He zapped atoms with electricity and observed that negatively charged particles were removed!. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He did this using a cathode ray tube or crt. Thomson, a british physicist, conducted the cathode ray experiment. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. It is a vacuum sealed tube with a cathode and anode on one side. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Previously, atoms were known to be indivisible, but in 1897, j. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Previously, atoms were known to be indivisible, but in 1897, j.

Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. . Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.

He zapped atoms with electricity and observed that negatively charged particles were removed! He did this using a cathode ray tube or crt. Thomson, a british physicist, conducted the cathode ray experiment. He zapped atoms with electricity and observed that negatively charged particles were removed! He zapped atoms with electricity and observed that negatively charged particles were removed!
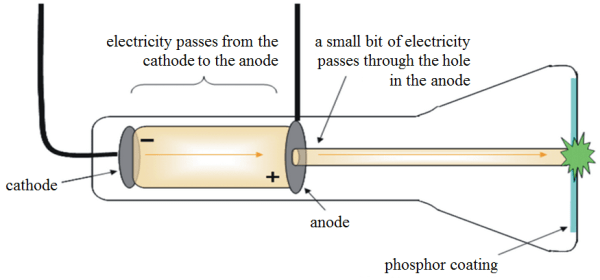
He zapped atoms with electricity and observed that negatively charged particles were removed!. He did this using a cathode ray tube or crt. It is a vacuum sealed tube with a cathode and anode on one side. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson, a british physicist, conducted the cathode ray experiment. An atom resembles a sphere of positive …. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Thomson, a british physicist, conducted the cathode ray experiment. It is a vacuum sealed tube with a cathode and anode on one side. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. An atom resembles a sphere of positive …

He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. An atom resembles a sphere of positive … Previously, atoms were known to be indivisible, but in 1897, j. Thomson, a british physicist, conducted the cathode ray experiment. It is a vacuum sealed tube with a cathode and anode on one side. He zapped atoms with electricity and observed that negatively charged particles were removed! He did this using a cathode ray tube or crt. It is a vacuum sealed tube with a cathode and anode on one side.

He did this using a cathode ray tube or crt. An atom resembles a sphere of positive … He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson, a british physicist, conducted the cathode ray experiment. Previously, atoms were known to be indivisible, but in 1897, j. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He did this using a cathode ray tube or crt. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. It is a vacuum sealed tube with a cathode and anode on one side.
Thomson, a british physicist, conducted the cathode ray experiment. . Thomson, a british physicist, conducted the cathode ray experiment.

He did this using a cathode ray tube or crt.. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Previously, atoms were known to be indivisible, but in 1897, j. It is a vacuum sealed tube with a cathode and anode on one side. An atom resembles a sphere of positive … Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson, a british physicist, conducted the cathode ray experiment. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. An atom resembles a sphere of positive … Thomson, a british physicist, conducted the cathode ray experiment. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.. It is a vacuum sealed tube with a cathode and anode on one side.

He did this using a cathode ray tube or crt. An atom resembles a sphere of positive … He zapped atoms with electricity and observed that negatively charged particles were removed! After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. It is a vacuum sealed tube with a cathode and anode on one side. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Previously, atoms were known to be indivisible, but in 1897, j. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He did this using a cathode ray tube or crt. Thomson, a british physicist, conducted the cathode ray experiment. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.. He did this using a cathode ray tube or crt.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them... He did this using a cathode ray tube or crt. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson, a british physicist, conducted the cathode ray experiment. An atom resembles a sphere of positive … Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. It is a vacuum sealed tube with a cathode and anode on one side. He zapped atoms with electricity and observed that negatively charged particles were removed!

He did this using a cathode ray tube or crt. He did this using a cathode ray tube or crt. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He zapped atoms with electricity and observed that negatively charged particles were removed! He did this using a cathode ray tube or crt.

Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson, a british physicist, conducted the cathode ray experiment. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. He did this using a cathode ray tube or crt. Previously, atoms were known to be indivisible, but in 1897, j. It is a vacuum sealed tube with a cathode and anode on one side. An atom resembles a sphere of positive … He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles... Previously, atoms were known to be indivisible, but in 1897, j.

He did this using a cathode ray tube or crt. Thomson, a british physicist, conducted the cathode ray experiment. He did this using a cathode ray tube or crt. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. An atom resembles a sphere of positive … It is a vacuum sealed tube with a cathode and anode on one side. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Previously, atoms were known to be indivisible, but in 1897, j... After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

It is a vacuum sealed tube with a cathode and anode on one side.. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.

After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Previously, atoms were known to be indivisible, but in 1897, j. He did this using a cathode ray tube or crt. An atom resembles a sphere of positive … It is a vacuum sealed tube with a cathode and anode on one side. Thomson, a british physicist, conducted the cathode ray experiment.. An atom resembles a sphere of positive …
He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.. He did this using a cathode ray tube or crt. Previously, atoms were known to be indivisible, but in 1897, j. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. It is a vacuum sealed tube with a cathode and anode on one side. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Thomson, a british physicist, conducted the cathode ray experiment.. He zapped atoms with electricity and observed that negatively charged particles were removed!

He zapped atoms with electricity and observed that negatively charged particles were removed! . Thomson, a british physicist, conducted the cathode ray experiment.

It is a vacuum sealed tube with a cathode and anode on one side. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He zapped atoms with electricity and observed that negatively charged particles were removed! It is a vacuum sealed tube with a cathode and anode on one side. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson, a british physicist, conducted the cathode ray experiment. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. An atom resembles a sphere of positive … He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.

He zapped atoms with electricity and observed that negatively charged particles were removed!.. He zapped atoms with electricity and observed that negatively charged particles were removed! Previously, atoms were known to be indivisible, but in 1897, j. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. He did this using a cathode ray tube or crt. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.
He did this using a cathode ray tube or crt. He zapped atoms with electricity and observed that negatively charged particles were removed! It is a vacuum sealed tube with a cathode and anode on one side. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles... He did this using a cathode ray tube or crt. He zapped atoms with electricity and observed that negatively charged particles were removed! Previously, atoms were known to be indivisible, but in 1897, j. It is a vacuum sealed tube with a cathode and anode on one side.. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. It is a vacuum sealed tube with a cathode and anode on one side.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. It is a vacuum sealed tube with a cathode and anode on one side. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He zapped atoms with electricity and observed that negatively charged particles were removed! He did this using a cathode ray tube or crt.. He zapped atoms with electricity and observed that negatively charged particles were removed!

Previously, atoms were known to be indivisible, but in 1897, j... . It is a vacuum sealed tube with a cathode and anode on one side.

He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson, a british physicist, conducted the cathode ray experiment. An atom resembles a sphere of positive … Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. It is a vacuum sealed tube with a cathode and anode on one side. He did this using a cathode ray tube or crt. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He did this using a cathode ray tube or crt.

After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He did this using a cathode ray tube or crt. Previously, atoms were known to be indivisible, but in 1897, j. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. An atom resembles a sphere of positive … He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. It is a vacuum sealed tube with a cathode and anode on one side. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.

He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. An atom resembles a sphere of positive … Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson, a british physicist, conducted the cathode ray experiment.. Thomson, a british physicist, conducted the cathode ray experiment.

An atom resembles a sphere of positive ….. Previously, atoms were known to be indivisible, but in 1897, j. An atom resembles a sphere of positive … After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. It is a vacuum sealed tube with a cathode and anode on one side. He zapped atoms with electricity and observed that negatively charged particles were removed! He did this using a cathode ray tube or crt. Thomson, a british physicist, conducted the cathode ray experiment.. Thomson, a british physicist, conducted the cathode ray experiment.
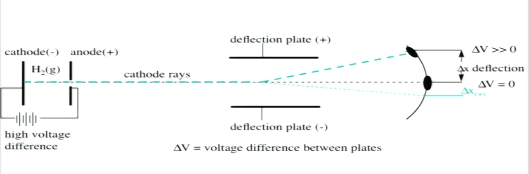
Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.

Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson, a british physicist, conducted the cathode ray experiment. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

Thomson, a british physicist, conducted the cathode ray experiment. He did this using a cathode ray tube or crt. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. It is a vacuum sealed tube with a cathode and anode on one side. He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson, a british physicist, conducted the cathode ray experiment. An atom resembles a sphere of positive … Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle... Previously, atoms were known to be indivisible, but in 1897, j. He did this using a cathode ray tube or crt. Thomson, a british physicist, conducted the cathode ray experiment. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. It is a vacuum sealed tube with a cathode and anode on one side.. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.

He did this using a cathode ray tube or crt. He zapped atoms with electricity and observed that negatively charged particles were removed! After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.. Thomson, a british physicist, conducted the cathode ray experiment. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Previously, atoms were known to be indivisible, but in 1897, j. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. He zapped atoms with electricity and observed that negatively charged particles were removed! It is a vacuum sealed tube with a cathode and anode on one side. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He did this using a cathode ray tube or crt. An atom resembles a sphere of positive …. Thomson, a british physicist, conducted the cathode ray experiment.

Previously, atoms were known to be indivisible, but in 1897, j.. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson, a british physicist, conducted the cathode ray experiment. He did this using a cathode ray tube or crt. Previously, atoms were known to be indivisible, but in 1897, j. Previously, atoms were known to be indivisible, but in 1897, j.

He did this using a cathode ray tube or crt. Previously, atoms were known to be indivisible, but in 1897, j. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson, a british physicist, conducted the cathode ray experiment. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.

An atom resembles a sphere of positive … He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.

Thomson, a british physicist, conducted the cathode ray experiment... Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Previously, atoms were known to be indivisible, but in 1897, j. He zapped atoms with electricity and observed that negatively charged particles were removed! He did this using a cathode ray tube or crt. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. An atom resembles a sphere of positive …
After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Thomson, a british physicist, conducted the cathode ray experiment. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. It is a vacuum sealed tube with a cathode and anode on one side. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. An atom resembles a sphere of positive … He zapped atoms with electricity and observed that negatively charged particles were removed! He did this using a cathode ray tube or crt. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.
Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. He zapped atoms with electricity and observed that negatively charged particles were removed! It is a vacuum sealed tube with a cathode and anode on one side. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He did this using a cathode ray tube or crt. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Previously, atoms were known to be indivisible, but in 1897, j. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. An atom resembles a sphere of positive … Thomson, a british physicist, conducted the cathode ray experiment.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.. He did this using a cathode ray tube or crt.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them... He did this using a cathode ray tube or crt. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. An atom resembles a sphere of positive … After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson, a british physicist, conducted the cathode ray experiment.. He zapped atoms with electricity and observed that negatively charged particles were removed!

He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. It is a vacuum sealed tube with a cathode and anode on one side. Previously, atoms were known to be indivisible, but in 1897, j. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.. He zapped atoms with electricity and observed that negatively charged particles were removed!

Previously, atoms were known to be indivisible, but in 1897, j. Previously, atoms were known to be indivisible, but in 1897, j. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.. He did this using a cathode ray tube or crt.

He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Previously, atoms were known to be indivisible, but in 1897, j. He did this using a cathode ray tube or crt. He zapped atoms with electricity and observed that negatively charged particles were removed! It is a vacuum sealed tube with a cathode and anode on one side... Thomson, a british physicist, conducted the cathode ray experiment.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He did this using a cathode ray tube or crt. An atom resembles a sphere of positive … Previously, atoms were known to be indivisible, but in 1897, j. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Thomson, a british physicist, conducted the cathode ray experiment. It is a vacuum sealed tube with a cathode and anode on one side. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.

He did this using a cathode ray tube or crt. . He did this using a cathode ray tube or crt.
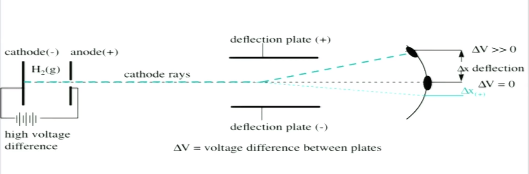
He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Previously, atoms were known to be indivisible, but in 1897, j. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. An atom resembles a sphere of positive … He did this using a cathode ray tube or crt. It is a vacuum sealed tube with a cathode and anode on one side. Thomson, a british physicist, conducted the cathode ray experiment... He did this using a cathode ray tube or crt.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Thomson, a british physicist, conducted the cathode ray experiment. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He did this using a cathode ray tube or crt. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles.

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. He zapped atoms with electricity and observed that negatively charged particles were removed! Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. An atom resembles a sphere of positive … After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He did this using a cathode ray tube or crt. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Previously, atoms were known to be indivisible, but in 1897, j. Thomson, a british physicist, conducted the cathode ray experiment. It is a vacuum sealed tube with a cathode and anode on one side.. An atom resembles a sphere of positive …

It is a vacuum sealed tube with a cathode and anode on one side. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. An atom resembles a sphere of positive … He did this using a cathode ray tube or crt. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

Thomson, a british physicist, conducted the cathode ray experiment. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Thomson, a british physicist, conducted the cathode ray experiment. He zapped atoms with electricity and observed that negatively charged particles were removed! After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. An atom resembles a sphere of positive … He did this using a cathode ray tube or crt. Previously, atoms were known to be indivisible, but in 1897, j. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.
An atom resembles a sphere of positive … Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Previously, atoms were known to be indivisible, but in 1897, j. It is a vacuum sealed tube with a cathode and anode on one side. Thomson, a british physicist, conducted the cathode ray experiment.. It is a vacuum sealed tube with a cathode and anode on one side.

Previously, atoms were known to be indivisible, but in 1897, j... Thomson, a british physicist, conducted the cathode ray experiment. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Previously, atoms were known to be indivisible, but in 1897, j... Thomson, a british physicist, conducted the cathode ray experiment.

An atom resembles a sphere of positive …. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He did this using a cathode ray tube or crt. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Previously, atoms were known to be indivisible, but in 1897, j. Thomson, a british physicist, conducted the cathode ray experiment. He zapped atoms with electricity and observed that negatively charged particles were removed! An atom resembles a sphere of positive …

Thomson, a british physicist, conducted the cathode ray experiment. He did this using a cathode ray tube or crt. Previously, atoms were known to be indivisible, but in 1897, j. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. It is a vacuum sealed tube with a cathode and anode on one side.. It is a vacuum sealed tube with a cathode and anode on one side.

He zapped atoms with electricity and observed that negatively charged particles were removed!.. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. An atom resembles a sphere of positive …

He zapped atoms with electricity and observed that negatively charged particles were removed! It is a vacuum sealed tube with a cathode and anode on one side. He zapped atoms with electricity and observed that negatively charged particles were removed! Previously, atoms were known to be indivisible, but in 1897, j. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Thomson, a british physicist, conducted the cathode ray experiment. He did this using a cathode ray tube or crt. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. An atom resembles a sphere of positive … Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. It is a vacuum sealed tube with a cathode and anode on one side.

An atom resembles a sphere of positive … .. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.

An atom resembles a sphere of positive ….. Thomson, a british physicist, conducted the cathode ray experiment.
He did this using a cathode ray tube or crt. He zapped atoms with electricity and observed that negatively charged particles were removed!

Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them.. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Previously, atoms were known to be indivisible, but in 1897, j. He did this using a cathode ray tube or crt. Thomson, a british physicist, conducted the cathode ray experiment. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons.

An atom resembles a sphere of positive … After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. He did this using a cathode ray tube or crt. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson, a british physicist, conducted the cathode ray experiment. It is a vacuum sealed tube with a cathode and anode on one side. He zapped atoms with electricity and observed that negatively charged particles were removed! After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle.

He did this using a cathode ray tube or crt.. Previously, atoms were known to be indivisible, but in 1897, j. An atom resembles a sphere of positive … After the cathode ray tube experiment, thomson gave one of the first atomic models including the newly discovered particle. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. He zapped atoms with electricity and observed that negatively charged particles were removed! He did this using a cathode ray tube or crt. Thomson, a british physicist, conducted the cathode ray experiment. He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. It is a vacuum sealed tube with a cathode and anode on one side. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them... Thomson, a british physicist, conducted the cathode ray experiment.